Get the Facts About COVID-19 Vaccines
The UFCW knows how hard our members and other essential workers continue to work to provide for their communities during the pandemic. We want to make sure our members have the important information they need about the COVID-19 vaccine and how to access it.
For the most up-to-date information and find Covid-19 vaccination sites near you, please visit vaccine.gov.
Recent Updates

How to prepare and protect yourself for flu season during the COVID-19 pandemic
Download Our COVID Vaccine Flyer for Your Workplace
Hear from Experts About the Importance of the COVID-19 Vaccine
Earlier this year, the UFCW hosted an educational COVID-19 Q&A Series with top doctors and leading public health specialists from across the country to answer your questions about the COVID-19 disease and the life-saving vaccines for it.
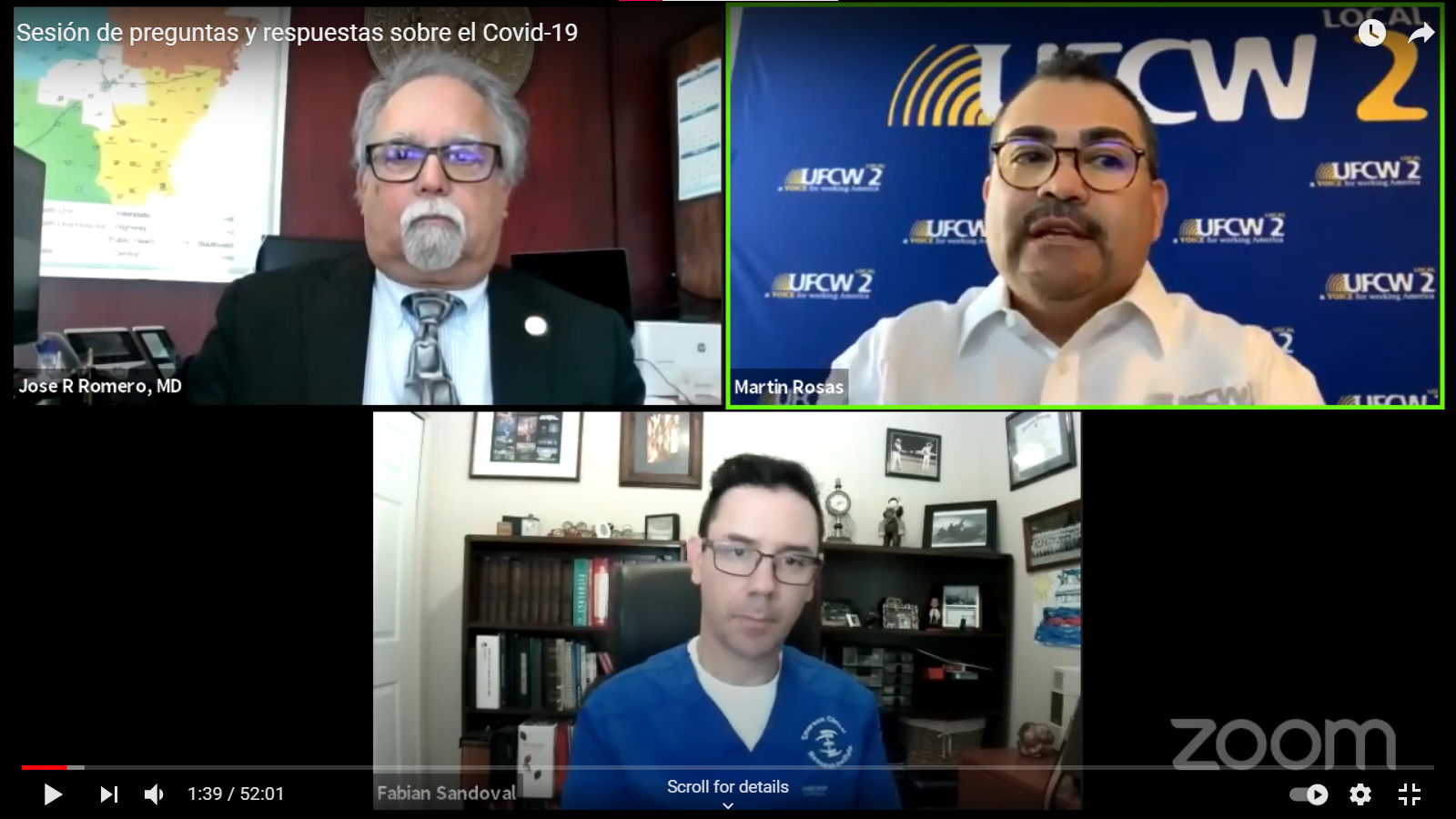
May 6 Q&A Series Town Hall [Video in Spanish]
Panelists
Dr. José Romero, MD
Chair at Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
Dr. Fabian Sandoval, MD
Executive Director and Research Director at the Emerson Clinical Research Institute.
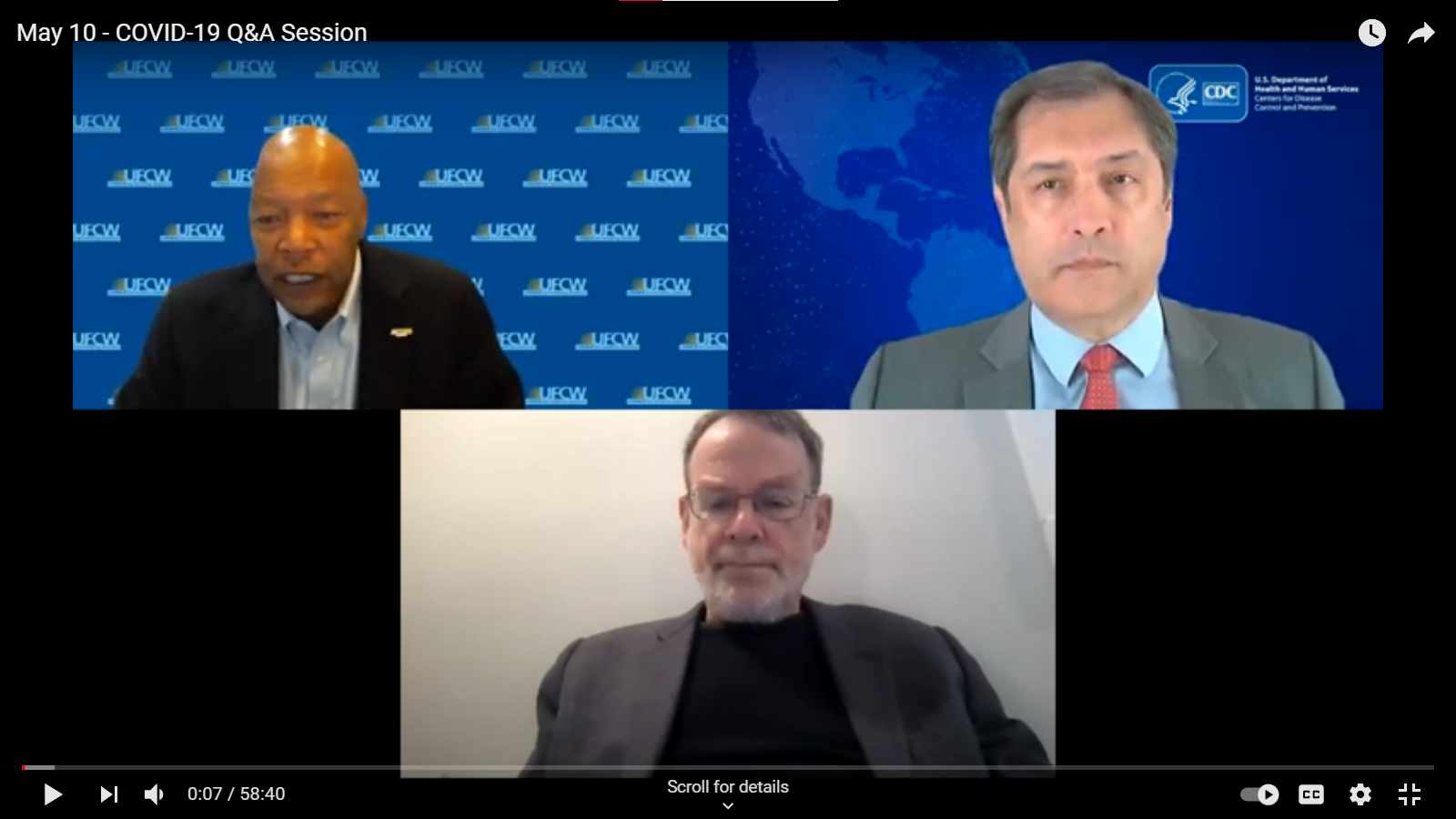
May 10 Q&A Series Town Hall
Panelists
Dr. Peter Marks, M.D., PhD
Director of the Center for Biologics Evaluation and Research at the Food and Drug Administration which is the agency responsible for regulating and reviewing the safety and efficacy of COVID-19 vaccine candidates.
Dr. José Montero, MD, MHCDS
Director of the Center for State, Tribal, Local, and Territorial Support at the CDC, where he oversees support to the US health departments and those serving tribal nations and insular areas.
Dr. Robert Harrison, MD, MPH
Clinical professor of medicine at UCSF and an occupational and environmental medicine specialist. He specializes in preventing coronavirus spread in the workplace environment.
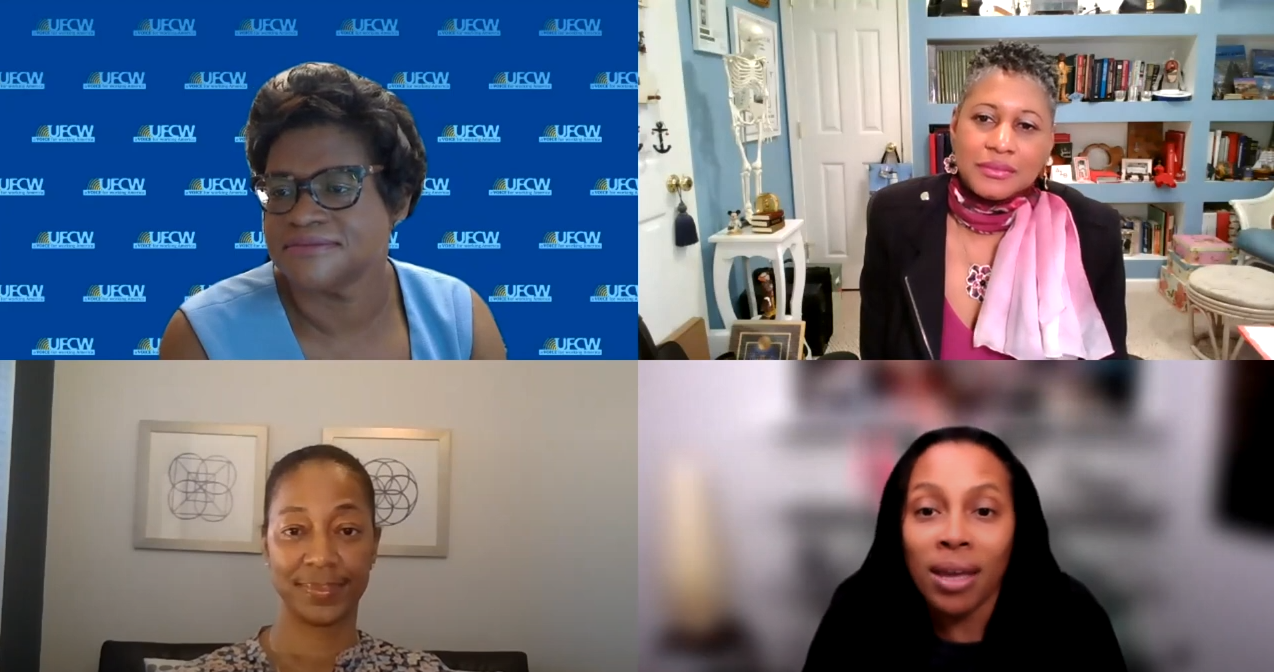
May 27 Q&A Series Town Hall
Panelists
Dr. Marcella Nunez Smith, MD, MHS
Senior Advisor, White House COVID-19 Response Team; Associate Professor of Internal Medicine, Public Health, and Management; Founding Director of the Equity Research and Innovation Center; Director of the Center for Research Engagement, Yale School of Medicine.
Dr. Sherita Hill Golden, MD, MHS
Vice President of the Hugh P. McCormick Family Professor of Endocrinology and Metabolism and Chief Diversity Officer at Johns Hopkins Medicine.
Ms. Tiffany Tate, MHS
Executive Director of the Maryland Partnership for Prevention, a non-profit organization that is the parent to Maryland’s childhood and adult immunization coalitions
Get The Facts Vaccine Flyer
Download and post the Get The Facts vaccine flyer (in English and Spanish) on your work bulletin board. Click on the image to download the PDF.